CLINICAL DIAGNOSTICS
Bringing proteomics to the clinic

CLINICAL DIAGNOSTICS
MRM Proteomics Inc. recognizes the promise of proteomics for clinical applications.
This vision has led us to partner on projects with PROOF Centre of Excellence and the Jain Foundation for developing proteomics-based biomarkers. The development of laboratory-developed tests (LDTs) will provide health-care providers with critical information to guide risk management, diagnosis and treatment plans. The value of biomarkers to the drug development pipeline is also well-established. Effective use of biomarkers in clinical trials can greatly expedite the process of bringing a drug to market. We are currently involved in a project with academic partners and AstraZeneca to develop and implement an iMALDI assay for patient stratification in clinical trials. See Press Releases for the details on these projects and others.

immunoMALDI (iMALDI)
MRM Proteomics Inc. holds a license for patented iMALDI technology that enables rapid, robust, highly automated protein quantitation in complex samples. The iMALDI approach involves spiking protein digest from a biological sample with a stable isotope-labeled standard peptide matching the target, after which the endogenous peptides and standard peptides are co-captured on antibodies conjugated to magnetic beads.
The peptides are eluted from the antibodies on a MALDI plate by applying acidic MALDI matrix, followed by MALDI-MS analysis. The use of mass spectrometry (MS) with internal standards ensures highly precise quantitation. Furthermore, detection with MS provides added protection against interferences, since the assay does not depend exclusively on the antibody for specificity. This method is compatible with the Bruker Biotyper instrument, which has already been granted FDA approval for applications in microbiology.
iMALDI FEATURES & BENEFITS

Robust & easy to use

Uses SIS peptides for reproducible, high precision quantitation

Does not depend on antibodies for specificity

Capable of isoform discrimination

High-throughput & compatible with automation

Compatible with equipment already installed in clinical labs

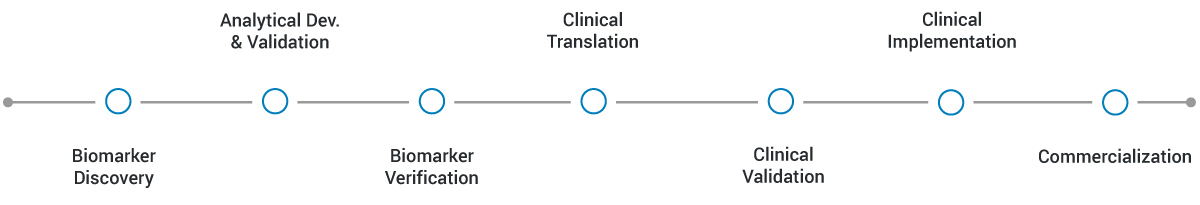
APPLICATIONS & CURRENT PROJECTS
MRM Proteomics Inc is already playing a key role in the development and validation of technologies for clinical proteomics.
Project examples are shown below.

ASSOCIATED REFERENCES
Sep 2018
Popp R, Li H, Borchers CH
Expert review of proteomics
Aug 2018
Zahedi RP, Parker CE, Borchers CH
Clinical chemistry
May 2018
Popp R, Basik M, Spatz A, Batist G, Zahedi RP, Borchers CH
Mar 2018
Parker CE, Borchers CH
PROTEOMICS – Clinical Applications
Jun 2015
Popp R, Malmström D, Chambers AG, Lin D, Camenzind AG, van der Gugten JG, Holmes DT, Pugia M, Jaremek M, Cornett S, Suckau D, Borchers CH
Biochimica et Biophysica Acta
Dec 2013
Camenzind AG, van der Gugten JG, Popp R, Holmes DT, Borchers CH
Feb 2012
Mason DR, Reid JD, Camenzind AG, Holmes DT, Borchers CH

 +1 (800) 559-9921
+1 (800) 559-9921